MedVasc presents successful study results at international conference
 Michael Åkesson, CEO and founder of MedVasc
Michael Åkesson, CEO and founder of MedVascMichael Åkesson, CEO and founder of the medtech company MedVasc, has been accepted as a speaker at the most prestigious international symposium of the year for researchers, the medical device industry and physicians specializing in cardiovascular diseases: the Charring Cross Symposium (CX), which will be held virtually April 19-22.
Dr. Michael Åkesson, a surgeon and researcher at Lund University Hospital, will present the extremely promising results obtained in a clinical trial in which his innovative medical device has been used to anesthetize patients prior to varicose vein surgery. He is further refining the innovation within his company, MedVasc.
About 30 percent of the population worldwide suffers from varicose veins. The anesthesia process is usually painful for patients undergoing treatment, but Dr. Michael Åkesson has developed an instrument that will reduce this problem. Once the instrument has been inserted into the varicose vein, local anesthesia is delivered from the inside, where there are no pain receptors, after which laser surgery is carried out.
The purpose of the study conducted using this instrument was to verify safety and function, as well as to assess pain management in conjunction with the MedVasc Solutio™ technique. The results were extremely encouraging: all treated patients reported minimal pain, and the surgeons performing the procedure considered the instrument to be safe and easy to use.
The study results have now been analyzed and a scientific article has been approved for presentation at the Charring Cross International Symposium in London, which will be held April 19-22, 2021. The CX Symposium is one of the largest scientific meetings in the world, where opinion leaders, doctors and medtech companies working with cardiovascular diseases meet and exchange ideas and results.
“We are extremely proud and pleased about the good function and safety demonstrated by our Solutio™ catheter; it more than met our expectations of providing essentially pain-free treatment to patients with varicose veins. Of course, more and larger studies are needed moving forward to confirm the advantages of our new technique,” says Dr. Michael Åkesson.
MedVasc is a member of SmiLe Incubator and a portfolio company under LU Holding, Lund University’s holding company.
For more information, please contact:
Michael Åkesson, CEO MedVasc AB, michael.akesson@medvasc.se; mobile +46705578131. www.medvasc.se
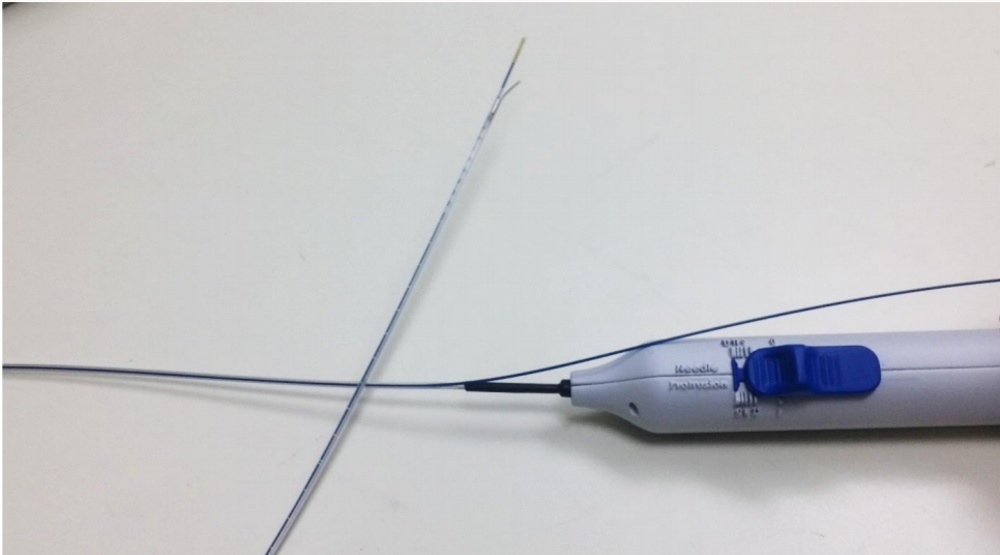
Image of the Solutio catheter showing the control handle with the blue slider that controls the needle at the other end of the catheter. Here the needle is in the extended position whereupon it deviates to the side, away from the blue laser fiber that is connected to the Solutio catheter. The bent needle deviates and penetrates the vessel wall so that the anesthetic can be applied outside the vessel through the needle.
About SmiLe Venture Hub – Pioneering Life Science and Foodtech Innovations
SmiLe Incubator is a life science business incubator based in Medicon Village in Lund. SmiLe helps entrepreneurs to commercialize their ideas. There are currently 25 companies in SmiLe, which together with alumni companies, have attracted more than EUR 380 million in venture capital to date since 2014. SmiLe offers business coaching, a large network of contacts and a dynamic community, as well as well-equipped laboratories which is unique of its kind in Sweden. SmiLe is a non-profit organization and receives basic funding from Region Skåne, Lund Municipality, Lund University and Medicon Village. SmiLe´s sponsors are Agilent, Sparbanken Skåne, Awa, Høiberg, Prevas, Setterwalls, Zacco, Aqilion. SmiLe’s listed alumni companies have a market capitalization of EUR 1,28 billion (Q3 2020). www.smileincubator.life